Empirical Formula of Butane
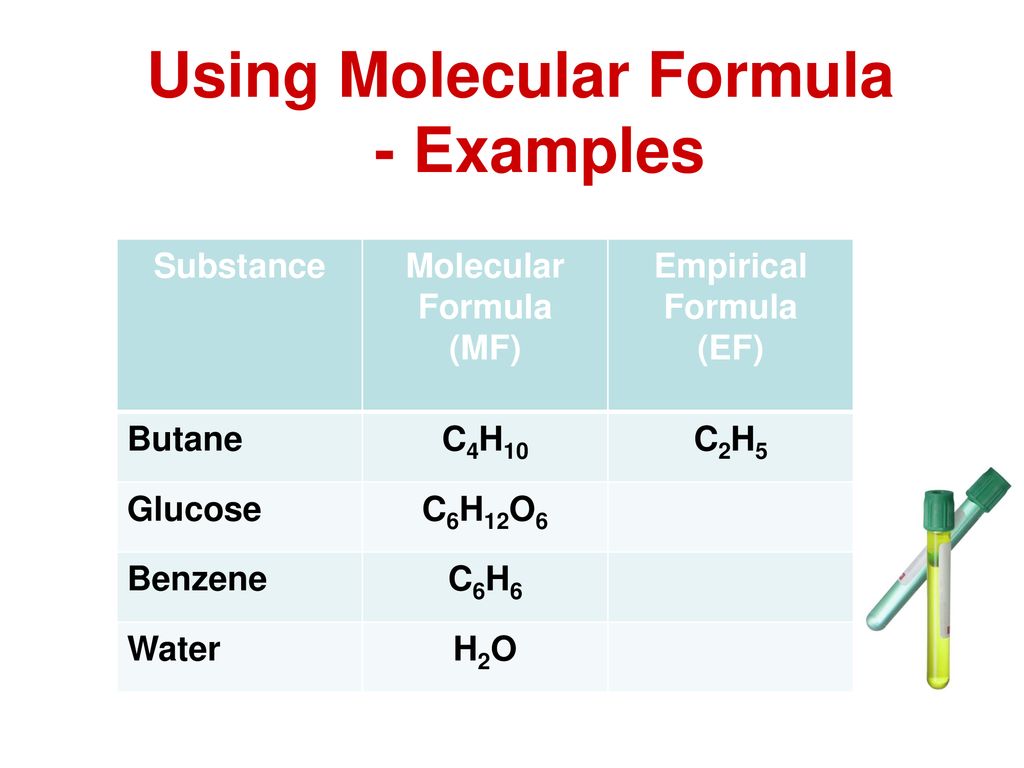
The molecular formula of butane is C 4 H 10. The molecular formula and empirical formula of some substances are the same.
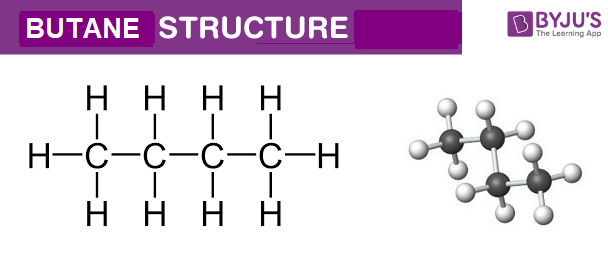
Butane C4h10 Structure Molecular Mass Properties Uses
C2H3 is the empirical formula for butane C4H6.

. The relative number of atoms of every element in the. This is the actual number of atoms of each element in a molecule of butane. But the measured molecular mass for Butane is given as 581224u.
This formula does not show. What is the empirical formula of butane. For every mole of carbon there are two moles of hydrogen.
This formula does not show. We can simplify the molecular formula C4 H10 which is the formula for butane by dividing the formula What best describes an. Find a molecular formula.
What is the empirical formula of butane. For example both types of formula for carbon. The empirical formula tells us the simplest whole-number ratio of the different types of atoms in a compound.
This is the actual number of atoms of each element in a molecule of butane. FROM PERCENTAGE COMPOSITION You can calculate the empirical formula from percentage composition. Join Login Class 10 General Knowledge Basic Science.
The empirical formula for C4 H10 is C2 H5. Click hereto get an answer to your question Calculate the empirical formula of butane. This is the actual number of atoms of each element in a molecule of butane.
FROM THE MOLECULAR FORMULA. What is the empirical formula for butane. By using the expression Molecular formula n empirical formula.
What is the empirical formula of butane. This gives the empirical formula of butane - C 2 H 5. The carbon-to-hydrogen ratio equals 23.
The molecular formula of butane is C 4 H 10. Hint Empirical formula is the simplest formula which provides the lowest whole number ratio of atoms which exist in the compound. This gives you the empirical formula CH.
The molecular formula of butane is C 4 H 10. What is the empirical formula for butane. C2H3 is the empirical formula for butane C4H6.
The carbon-to-hydrogen ratio equals 23. Solve Study Textbooks Guides. If you know that the molecular formula of.
For every mole of carbon there are two moles of hydrogen. This formula does not show.

Chem C10 Empirical Molecular Formula Flashcards Quizlet

Butane Molecular Geometry Hybridization Molecular Weight Molecular Formula Cas Number Bond Pairs Lone Pairs Lewis Structure

Butane Formula Structure What Is Butane Used For Video Lesson Transcript Study Com
No comments for "Empirical Formula of Butane"
Post a Comment